Oligodendroglial fatty acid metabolism as a central nervous system energy reserve
Other (20)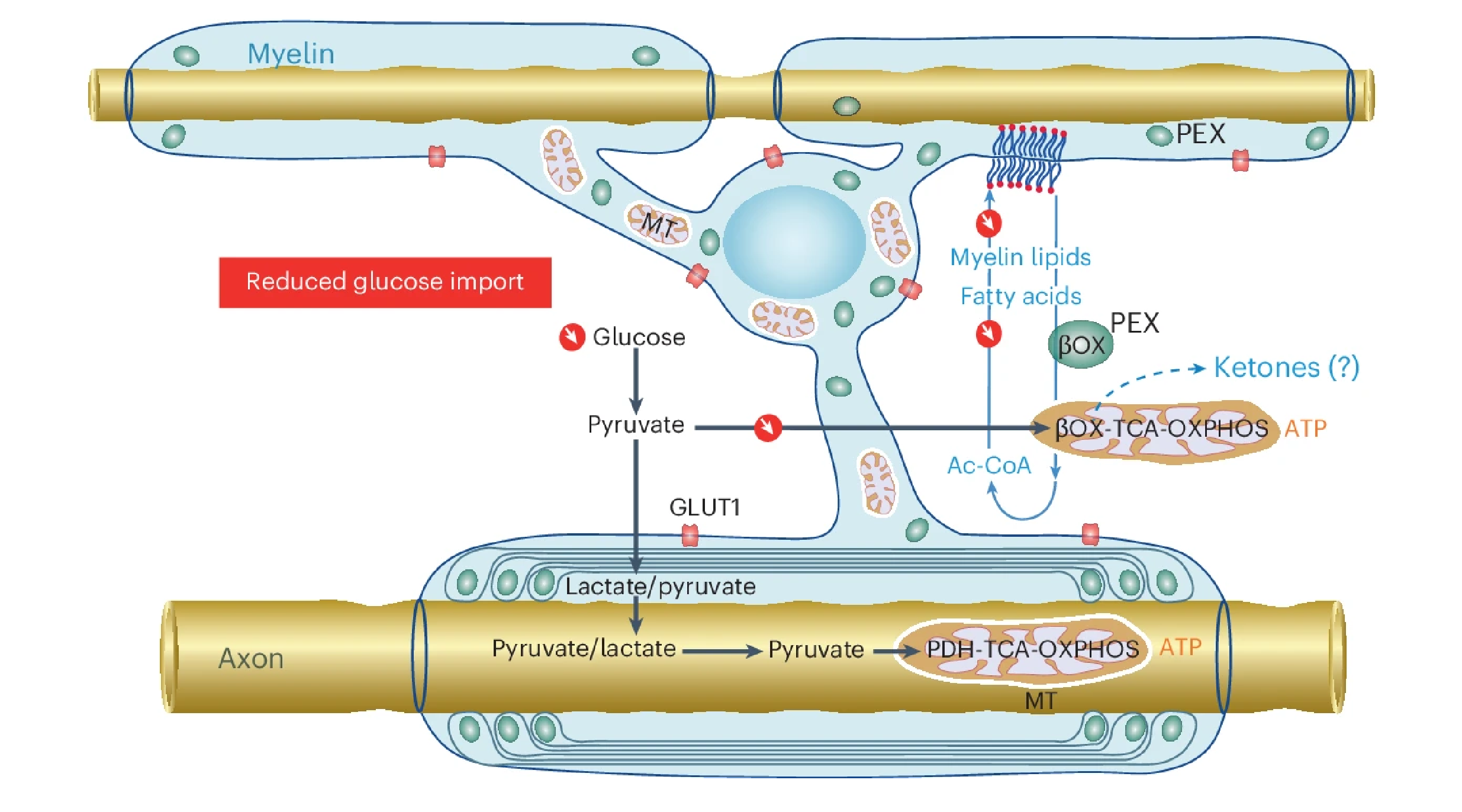
A recent study published in Nature Neuroscience reveals that oligodendrocytes, the myelinating cells of the central nervous system (CNS), play a crucial role in energy metabolism by utilizing fatty acids to support white matter function. This continuous lipid metabolism provides an energy reserve essential for maintaining axonal health, especially during glucose scarcity.
Key Findings:
- Enhanced Survival Under Glucose Deprivation: Oligodendrocytes demonstrate greater resilience to glucose deprivation compared to astrocytes, highlighting their capacity to adapt to energy shortages by metabolizing fatty acids.
- Fatty Acid β-Oxidation Dependency: Under low glucose conditions, both axonal ATP levels and action potentials become reliant on fatty acid β-oxidation, emphasizing the importance of lipid metabolism in sustaining neuronal activity.
- Protection Against Conduction Blocks: Fatty acid metabolism in oligodendrocytes safeguards axons from conduction blocks during glucose limitation, although it does not support high-frequency neuronal firing.
- Impact of GLUT1 Disruption: Reducing GLUT1 expression in oligodendrocytes disrupts myelin homeostasis, leading to gradual demyelination without overt behavioral changes, suggesting a delicate balance between myelin synthesis and degradation.
Implications:
These findings underscore the pivotal role of oligodendroglial lipid metabolism in CNS energy homeostasis. The ability of oligodendrocytes to utilize fatty acids as an energy source is vital for supporting axonal function, particularly when glucose is limited. Disruptions in this metabolic pathway can lead to myelin degradation, offering insights into neurological conditions characterized by demyelination.
For a comprehensive understanding, you can access the full article here: Oligodendroglial fatty acid metabolism as a central nervous system energy reserve.
Klaus-Armin Nave and Hannelore Ehrenreich are project leaders of TRR 274 project C01.





